When I told Rachel that I would write something for the favourite chemical reactions #chemcarnival being organized by C&EN, I started thinking about the reactions I did during my career at the bench (undergrad, PhD, and postdoc).
My least favourite reaction sprang to mind pretty quickly — but that’s a whole different blog post for the future. Let’s just say that I was working in a lab over the summer between my 2nd and 3rd years as an undergraduate, and the reaction involved a LOT of sodium cyanide and one of those coffee-bean grinders with the really sharp blades. Let your minds run free…
Having spent two years in the Mecca of metathesis at Caltech, it would have been too easy to choose that particular Nobel Prize-winning reaction as my favourite. The pull of ruthenium is strong — even though I joined Bob’s group to work on Mo-based chain-transfer agents, it wasn’t long before I was playing with the purple powder. The fact that the first Mo compound I made in Pasadena decomposed under vacuum (yes, I said vacuum — all I was doing was trying to dry it!), probably didn’t help.
So, my favourite reaction? Making and breaking imines. Synthetic chemistry is not just about making bonds, it’s about breaking them too you know.
Let’s start with the easy bit. Making an imine is a pretty simple process. Take an aldehyde and a primary amine and mix them together. You can add an acid catalyst if you want (not too strong mind, you don’t want to protonate your amine out of the equation). The amine attacks the carbonyl of the aldehyde and you generate a hemiaminal. These things don’t stick around for long because they eliminate water to give the imine (but if you’re really clever you can take a sneaky picture of one of them at low temperature in a well-defined solid).
The reaction exists as an equilibrium — the water can attack the imine and the sequence is reversed to give the aldehyde and the amine that you started with. What use is that you might ask? More on that later. Anyway, because we have an equilibrium, we can call on our friend Le Chatelier to help us out. If we can find a way to remove water from the system, the reaction will be driven to the right and we can make the imine in really high yields.
One particularly elegant way to remove the water is to use one of my favourite pieces of chemistry glassware, a Dean-Stark trap. You put one of these between your reaction flask and the condenser and you heat your aldehyde and amine in a solvent such as toluene that can form an azeotrope (one of my favourite chemistry words – see it’s all good!) with water. As the solvent condenses, it drops into the Dean-Stark trap and the water separates from the toluene and falls to the bottom of the trap because of its higher density — the only thing that gets recycled back into the reaction flask is toluene (assuming your trap is big enough to cope with the volume of water formed during the reaction). It’s just so neat and tidy.
Of course, not every imine you make will be particularly happy in boiling toluene, so there are other methods of removing the water, such as adding drying agents including magnesium sulfate or molecular sieves.
So, making imines is fairly straightforward. But why would we want to break them apart again? Well, imine formation (and cleavage) is an example of dynamic covalent chemistry, which can be really quite useful if you want to make just one particular product in a reaction that could potentially yield a vast number of different compounds. The idea is that if the reactants combine to form the incorrect product, that compound can be recycled back into the equilibrating reaction mixture and the system keeps evolving until it reaches an energy minimum. As such, the outcome of the reaction is under thermodynamic control (the final products are decided by their relative energies and the covalent bond-forming reactions are reversible) rather than kinetic control (the final products are decided by which reactions happen the quickest and the covalent bond-forming reactions are irreversible).
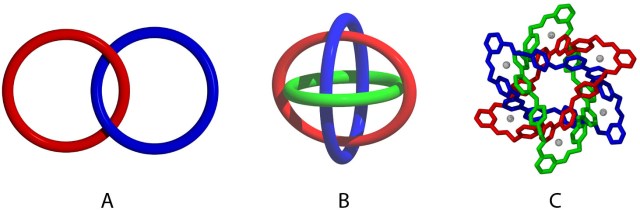
For example, if you react together a compound containing two aldehydes with one containing two amines, you could make lots of imine polymers of different lengths — as well as cyclic compounds where the ends of the polymer chains react intramolecularly. If imine formation was irreversible, you’d likely end up with a mess, but the reaction is reversible, so there exists the potential to bias the reaction to give a single product — the one that is energetically most stable. And that’s how you can make funky molecular topologies, such as the Borromean rings shown below (see this post for more background).
Dynamic covalent chemistry can also be applied in combinatorial systems — so-called dynamic combinatorial chemistry, pioneered by the likes of Jeremy Sanders and Jean-Marie Lehn (amongst others). Such approaches can be used to create synthetic hosts to bind small molecules, or conversely, to find small molecules that can bind to larger structures, such as inhibitors for enzymes as one example (subscription required).
Imines are just one example of a functional group that lends itself to this sort of dynamic chemistry. Others include esters, disulfides, hydrazones and even olefins (metathesis is a reversible reaction under the appropriate conditions). But imines are the ones I’ve played with myself, and so they’re my favourite.





Insight story
And breathe.
Closing issues of Nature Chemistry can sometimes be a bit frantic – and the just-published September issue was a little more of a challenge than usual. In addition to the usual types of content (research articles, reviews, research highlights, news & views pieces, and so on), this one contains a collection of Commentary articles that look at broader issues in chemistry beyond the science itself (I’ve written that last phrase so many times in the last few months… and I’m getting a bit sick of it to be honest).
The seeds of this special issue were sown well over a year ago in a meeting with my then boss (he’s climbed higher up the ladder since then). He asked what Nature Chemistry was going to do to mark the International Year of Chemistry. And the best answer I could come up with was something along the lines of, ‘erm… nothing… yet…’. After some discussion we decided it was an opportunity to do something special, so off I went to the rest of the team to try and figure out exactly what that would be.
There was really no point in doing more of what we do already. We routinely publish some pretty interesting research papers and review articles (or at least we think they’re interesting), and just upping the volume and slapping an IYC logo on some of them didn’t seem particularly satisfying. So, we decided that we’d get people to write opinion pieces on issues not directly related to science itself, but on broader issues related to the chemistry community, such as gender, education, communication, careers, sustainability, the developing world, and the future of pharma (OK, that last one is pretty close to the science, but cut us some slack!).
I just listed seven topics and you will see that we ended up with seven commentaries. We did actually commission eight articles, but one of them fell through. The less said about that, the better. Most, if not all, of the Commentaries were commissioned before the calendar flipped over into 2011 and first drafts started arriving in spring 2011. Over the summer, articles were edited and iterations were passed back and forth between the authors and the editorial team.
As our deadlines loomed (an August 23rd go-live date meant a press day – the day we finalise everything and send it off to the presses – of August 9th) we had to tidy up all the loose ends, such as making sure we had permissions to use all the images we wanted to and that we had the right credit information for them. These are the details that can make an editor’s head spin. Nevertheless, after a lot of hard work from our authors, from the editorial team, and last-but-not-least from our production team (art editor, copy editor, production editor, and web-production editor), these articles made it onto the printed page and onto the web. And this was in addition to the rest of the September issue that we had to put together as usual!
If you’re interested in chemistry and its future, I would recommend (but I am a bit biased I guess) going and reading these Commentaries – they are free until the end of September 2011. Matt has already blogged about his Commentary – all about communicating chemistry – and Michelle has also written a post about hers. And for the story behind the amazing mosaic cover of the September issue, head on over to the blog post I put up at the Sceptical Chymist.