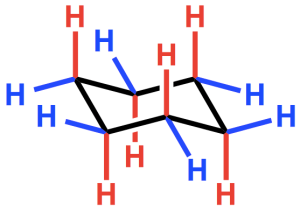
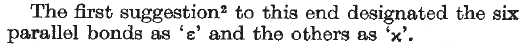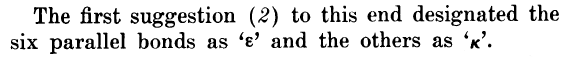Cyclohexane is undoubtedly an iconic molecule. Many of us learned to draw it (with varying degrees of proficiency) very early on in our organic chemistry classes as we were introduced to chairs, boats, half-chairs, twist-boats, cis, trans, A-values, conformation and, of course, axial and equatorial. Cyclohexane has six equatorial C–H bonds around the circumference of the ring and six axial C–H bonds, three pointing up and three pointing down.
It turns out that it wasn’t always ‘axial’ and ‘equatorial’ though… and I only discovered this last week when I came across a 1953 letter-to-the-editor in Nature (Nomenclature of cyclohexane bonds) from Barton, Hassel, Pitzer & Prelog.
The letter notes (shown below) that the labels first suggested for the different C–H bonds on the cyclohexane ring were ɛ (epsilon) for what we now call axial, and χ (chi) for what we refer to as equatorial these days:
The citation (the subscript ‘2’ in the excerpt above) is to a paper by Odd Hassel published in 1943 in something called Tidsskr. Kjemi, Bergv. Met. which turns out to be the Norwegian journal Tidsskrift For Kjemi Bergvesen og Metallurgi (I’m sure that’s cleared it up for you…). Even if my Norwegian was up to scratch (it isn’t), I thought it would be somewhat tricky to track down a copy of this article to find out the reasoning behind the choice of those particular descriptors.
Fortunately, the article was later translated into English and published in Topics in Stereochemistry in 1971, along with an article by Derek Barton that had originally been published in 1950 in the journal Experientia.
Hassel’s paper — The cyclohexane problem — explains the origin of the descriptors as follows:
For those of you paying attention, you may have noticed a problem. Whereas the Nature paper refers to ɛ and χ, the original Hassel paper refers to ɛ and κ (kappa). And based on the Greek origins of the descriptors, it is clear that it should be κ and not χ. Go back and look at the excerpt from the Nature paper above — even squinting a bit, it would need to be a very charitable interpretation to say that the symbol in question is κ and not χ. It seems that something went awry in the publication process (a couple of book chapters also confirm that the original descriptors were ɛ and κ).
One of these book chapters also pointed me in the direction of a 1954 Science paper, which shared the same authors (Barton, Hassel, Pitzer and Prelog) — and the same title — as the earlier 1953 Nature paper. On closer inspection, the letter in Science is, with one important exception, exactly the same as the one that appeared in Nature. See if you can spot the difference in this excerpt from the Science paper:
So, Science got it right; a kappa (κ) and not a chi (χ) for the equatorial bonds. Perhaps more remarkable, however, is that both Science and Nature published the *same* letter (the Nature letter was published on Dec 12, 1953 and the one in Science on Jan 1, 1954) — I wonder whether the editors knew of the dual publication…? Anyway, what was the ultimate purpose of this letter, this letter that was deemed so important that it should be published in both Science and Nature? Well, it was essentially just a proposal of new nomenclature for the different C–H bonds in cyclohexane.
After noting that the epsilon/kappa descriptors were difficult to remember, the authors pointed out that alternative nomenclature had been suggested by Beckett, Pitzer and Spitzer in a 1947 JACS paper. Basing their terms on geography rather than Greek, they had suggested the now-familiar ‘equatorial’ (e) for the C–H bonds around the equator of the ring and ‘polar’ (p) for the C–H bonds pointing either north or south away from the mean plane of the ring.
As highlighted in the Barton/Hassel/Pitzer/Prelog letter, however, the word ‘polar’ has another — very different — meaning in chemistry, and in an effort to prevent any confusion, they suggested that instead of polar, a better term would be ‘axial’. In another twist, the proposal to use ‘axial’ was actually made by Christopher Ingold who, despite this contribution, is only acknowledged in the Nature/Science letter (see below), rather than sharing in the authorship.
Considering that ‘equatorial’ was already suggested in the earlier Beckett, Pitzer and Spitzer JACS paper and ‘axial’ is Ingold’s idea, the role of Barton, Hassel and Prelog appears to be one of making an authoritative plea (with Pitzer) to the community for a new standard to be adopted, rather than defining the new nomenclature themselves.
UPDATE 11/05/2015 – here’s an interesting post about Hermann Sachse and his attempts to get his ideas about the conformation of cyclohexane across to the wider chemistry community towards the end of the 19th century.